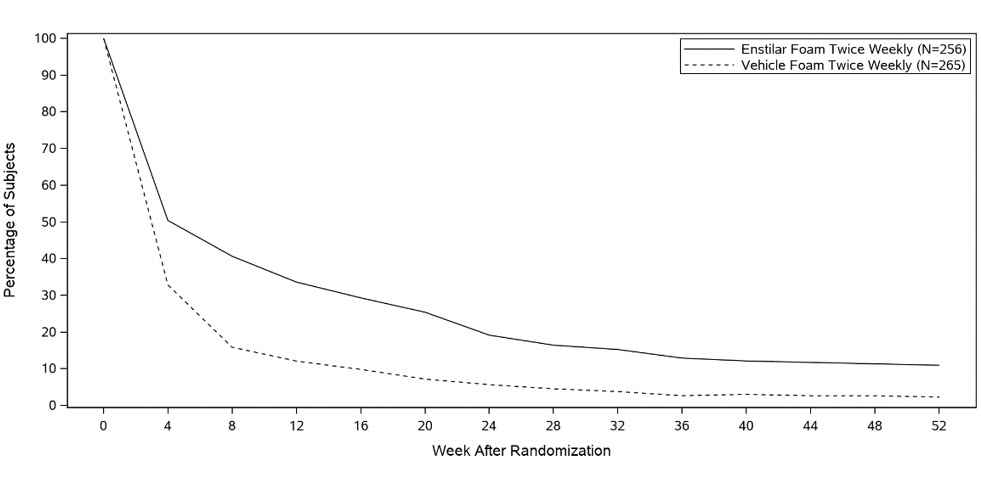
LEO Pharma Inc. Announces U.S. Food and Drug Administration (FDA) Expanded Regulatory Approvals for Enstilar® Foam and Taclonex® Topical Suspension in Treatment of Plaque Psoriasis | Business Wire

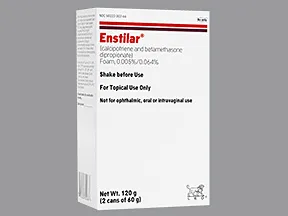
Enstilar (calcipatriene and betamethasone dipropionate) Foam 0.0005%/0.064%, 60 g Leo Pharma (RX) Ingredients and Reviews